
Hiteks, a trusted partner of physicians and FDA processes[1], announces its support of access to investigational products for patients with cancer by providing comprehensive services to oncology healthcare professionals in completing expanded access requests.
Key Features of Hiteks’ Expanded Access Program:
- Eases physician workflow related to the FDA application requirements for expanded access to investigational medical products.
- Supports thesystematic collection of data (i.e. FDA Form 3926)and streamlines data sharing with appropriate stakeholders (i.e. FDA, BioPharma Sponsor, and IRB).
- Reduces paperwork and bureaucracy by electronically storing and filing relevant data to support healthcare professionals in completing expanded access requests and downstream reporting:
- a. Seek and obtain LOA (Letter of Authorization) and product from BioPharma sponsor to ensure compliance with Expanded Access program
- b. Submit package to FDA: FDA 3926, CV,& LOA
- c. Submit annual reports on patient status to FDA
- d. Withdrawal IND, if appropriate
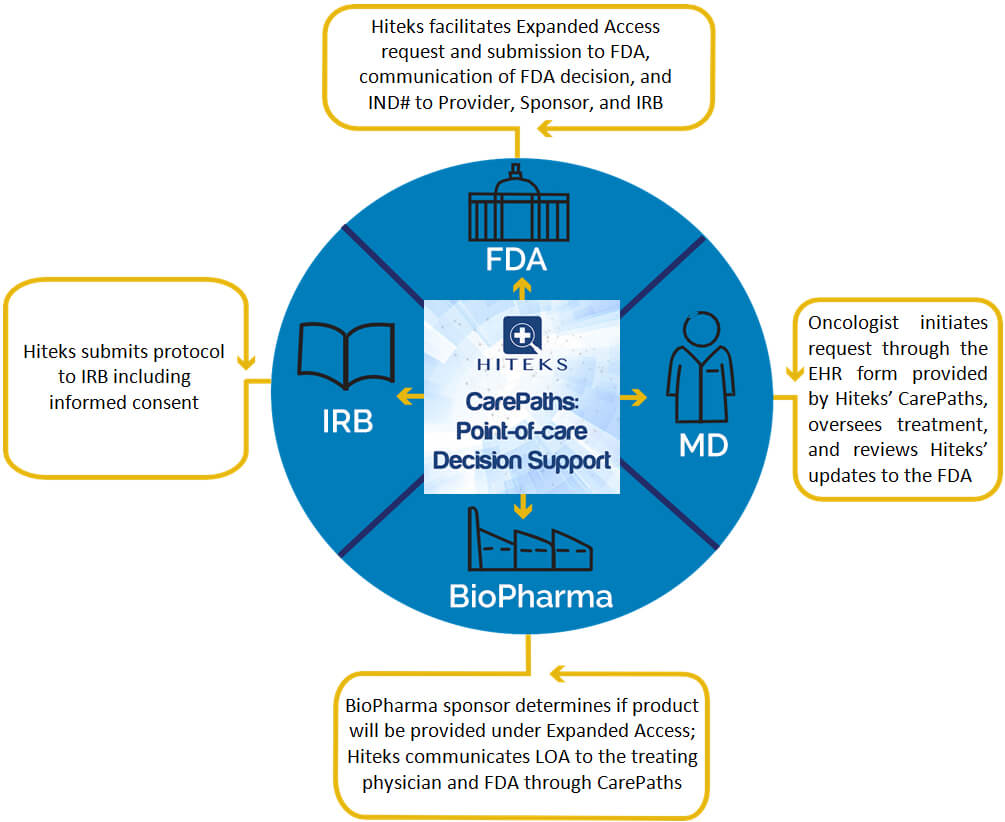
- Because Hiteks occupies a central place in the EHR workflow for providers, it is able to efficiently present options to providers who wish to complete the submission of a request for a Compassionate Use therapeutic.
- Hiteks retains a HIPAA-compliant database of information that can be used for downstream outcomes analysis and reporting.
Workflow supported by Hiteks’ CarePaths:
Expanded Access Decision Support
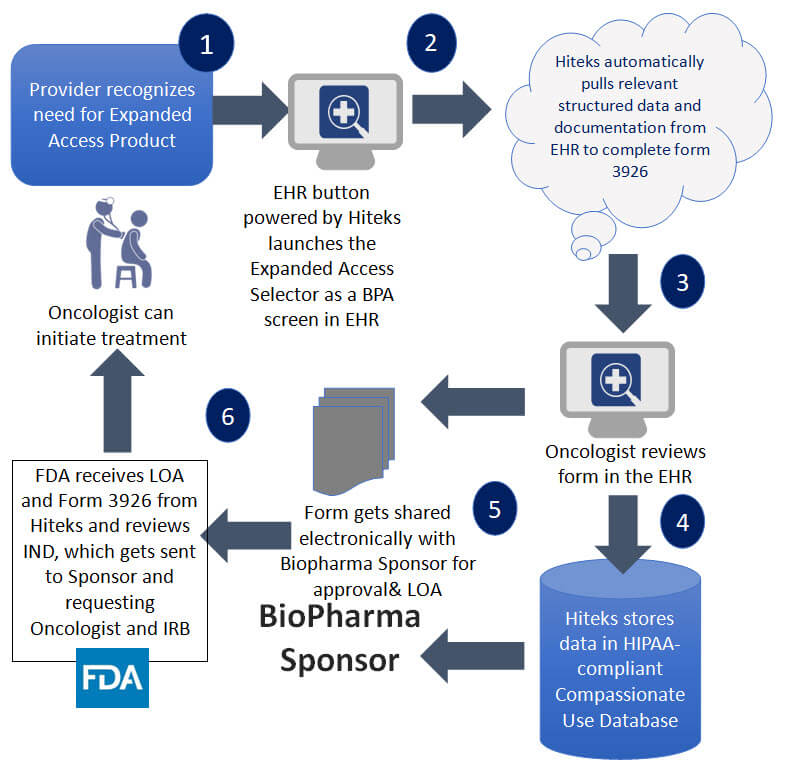
Form FDA 3926
Access to Hiteks’ CarePaths: Expanded Access for Epic 2019 Customers
6-weeks of elapsed time
- Clinical and Compliance Departments decide to use Hiteks for Expanded Access service
- Communication to Hiteks and Epic to begin implementation of solution integrated with Epic
- Hiteks coordinates with provider organization’s compliance and IT functions to configure solution within provider workflow according to workflow triggers specified in Epic’s External Decision Support framework via FHIR-based resources and Dynamic BPAs
- Provider IT downloads App Orchard credentials, Hiteks assigns provider ID and password, and Epic Test Server connects to Hiteks
- Provider validates the Expanded Access workflow functionality
- Physician and administrator training conducted
- Provider IT function copies the Epic Test configuration into Production& provider network team implements Epic Production rules for FHIR access
- System Go-Live

Gerry Petratos, MD, MS, CEO, Hiteks Solutions Inc.
Gerry has worked previously as Global Head of Healthcare Data Analytics at Roche and Genentech Pharmaceuticals before co-founding Hiteks in 2011. He trained as an NIH Fellow in Medical Informatics at the University of Utah and Intermountain Healthcare. He currently leads Hiteks’ decision support integration for CDI, oncology, cardiology, and infectious disease providers within EHR workflows.
About Hiteks:
- • Hiteks Solutions is privately held, physician-owned and managed organization
- • Founded in 2011 with corporate offices in New York City
- • Currently over 100 hospital and medical practice clients in 16 different States
- • Integrated with Epic since 2014

[1] Hiteks currently supports the Epic-integrated CarePaths: Point-of-Care Decision Support tool, available at https://apporchard.epic.com/Gallery?id=2724, and has previous experience working with the FDA under previous CRADA agreements, including for reporting and analysis of Adverse Events using Hiteks’ software.



